
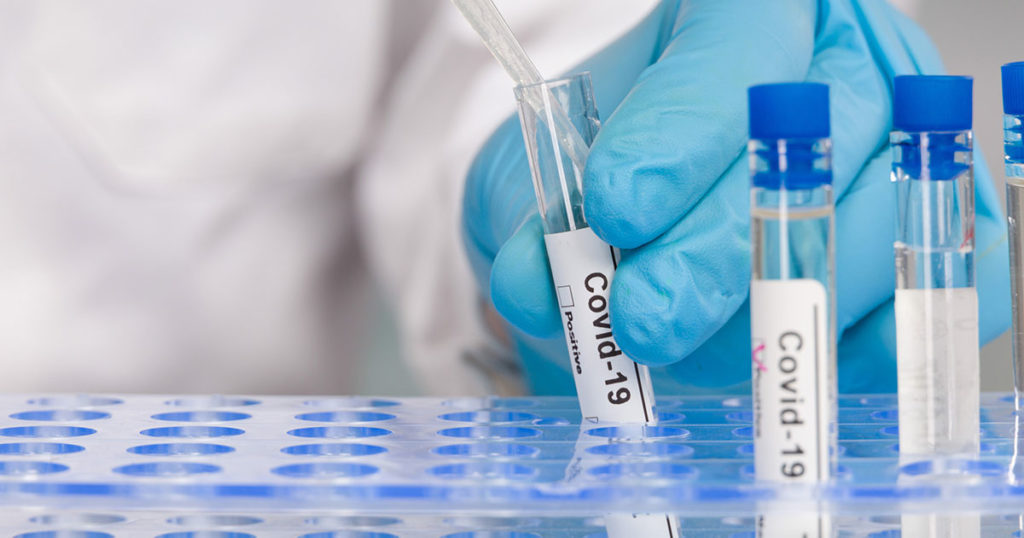
COVID-19: The Role Of In Vitro Diagnostic Testing – Backgrounder
Read background information on how AdvaMedDx®’s members produce IVD tests to detect COVID-19.

Read background information on how AdvaMedDx®’s members produce IVD tests to detect COVID-19.


Unequal access to medical technologies and, more broadly, health care services continues to persist – often disproportionately impacting the lives of racial and ethnic minorities.
AdvaMed® announced that shipments of COVID-19 molecular diagnostic tests reached one million per day for the week ending July 24, based on the latest data from the association’s National Testing Registry.
WASHINGTON, D.C. – The Advanced Medical Technology Association® (AdvaMed®), the Association of periOperative Registered Nurses (AORN), and the American Hospital Association (AHA) today announced 26 additional health care professional societies and…
AdvaMed®, the Association of periOperative Registered Nurses (AORN), and the American Hospital Association (AHA) today released “Re-entry Guidance for Health Care Facilities and Medical Device Representatives.”
Doctors, nurses and health care providers fighting on the front lines against the coronavirus are modern-day heroes. Every day they selflessly risk their own health to provide essential care to…

The Chinese government announced a process to export particular devices without NMPA approval.
View a flow chart of the evaluation process.

Foreign manufacturers of COVID-19 products with FDA approval can get on China’s export white list.