
Medtech Compliance Is More Complex so We Built a Program to Match
The AdvaMed Medtech Compliance Workshop is a forum designed specifically for medtech compliance leaders and legal and other professionals with health care compliance responsibilities.
AdvaMed®’s Medtech POV blog features all the latest developments, perspectives, and resources by and for the medical technology community.

The AdvaMed Medtech Compliance Workshop is a forum designed specifically for medtech compliance leaders and legal and other professionals with health care compliance responsibilities.

The MedTech Conference, brings together the companies shaping the future of innovation. Hosted by AdvaMed®, The Medtech Association, #MTC26 gives companies a focused moment to connect, collaborate, and turn conversations…

Early Alzheimer’s blood tests are improving diagnosis, but access depends on policy. Learn how expanded Medicare coverage is changing patient outcomes.

After the Supreme Court struck down IEEPA tariffs, CBP launched the CAPE system to process refunds. Here’s what importers need to know about eligibility, submission, and timing.
Brought to you by AdvaMed®, Medtech POV with Scott Whitaker is a podcast that covers the intersection of medtech and policy from every perspective.

The United States faces a maternal health crisis demanding urgent action. The U.S. has the worst maternal outcomes among high income nations. Our maternal mortality rates far exceed peer countries’,…

“The Insight Series: AI & Digital Health” features industry experts answering key questions to help inform policymakers and the public about how AI is shaping the future of care delivery.…
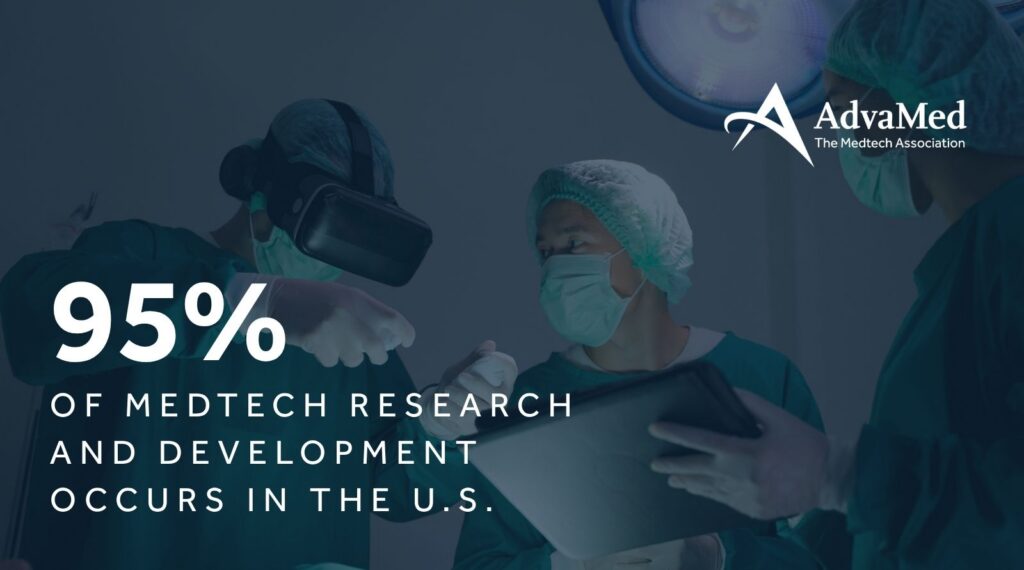
The tax incentives in the One Big Beautiful Bill Act passed by Congress and signed into law by President Trump last year are a prime example of the public policies reinforcing the…

“The Insight Series: AI & Digital Health” features industry experts answering key questions to help inform policymakers and the public about how AI is shaping the future of care delivery.…
If you have media inquiries, please contact:
Jim Jeffries, Senior Vice President, Public Affairs, 202-434-7242

AdvaMed® highlights its annual Awards program recognizing leaders whose careers have advanced innovation, patient access, and excellence across the medtech ecosystem.
Join over 500 medical technology companies as a member of one of the most effective trade associations in Washington, D.C.